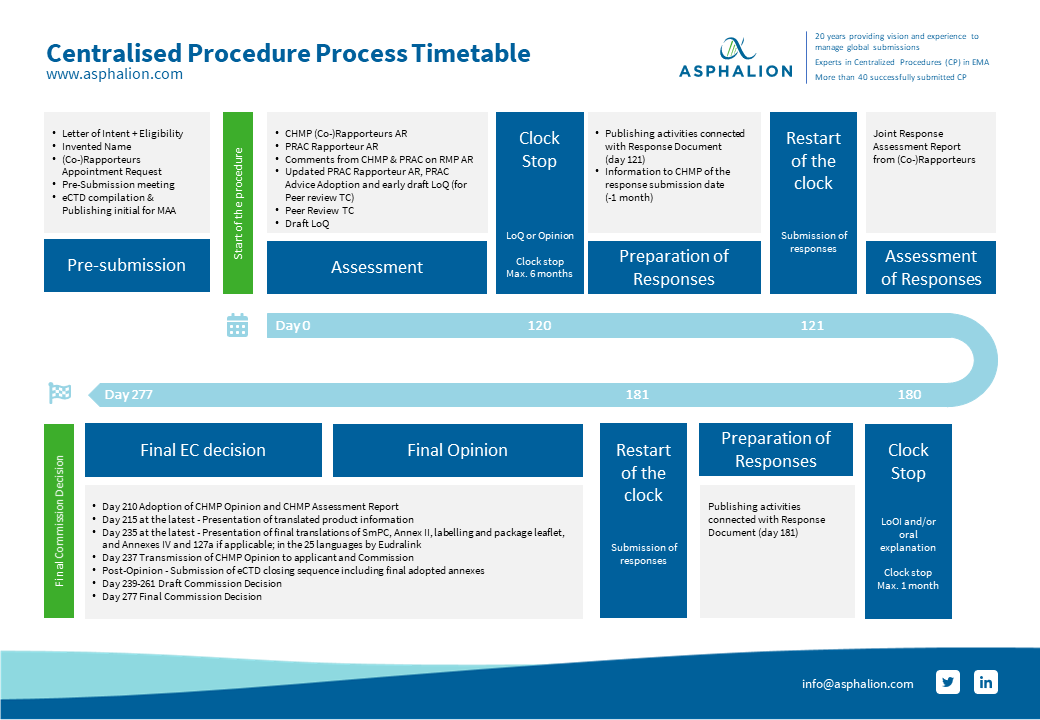
Comparison of regulatory pathways for the approval of advanced therapies in the European Union and the United States - Cytotherapy

Institutionnalisation du médiateur européen et nouveaux instruments d'enquête : le cas de l'enquête de 2017-2019 sur l'agence européenne des médicaments | Cairn.info
The European Medicines Agency (EMA) grants positive opinion for subcutaneous daratumumab in frontline and relapsed/refractory se
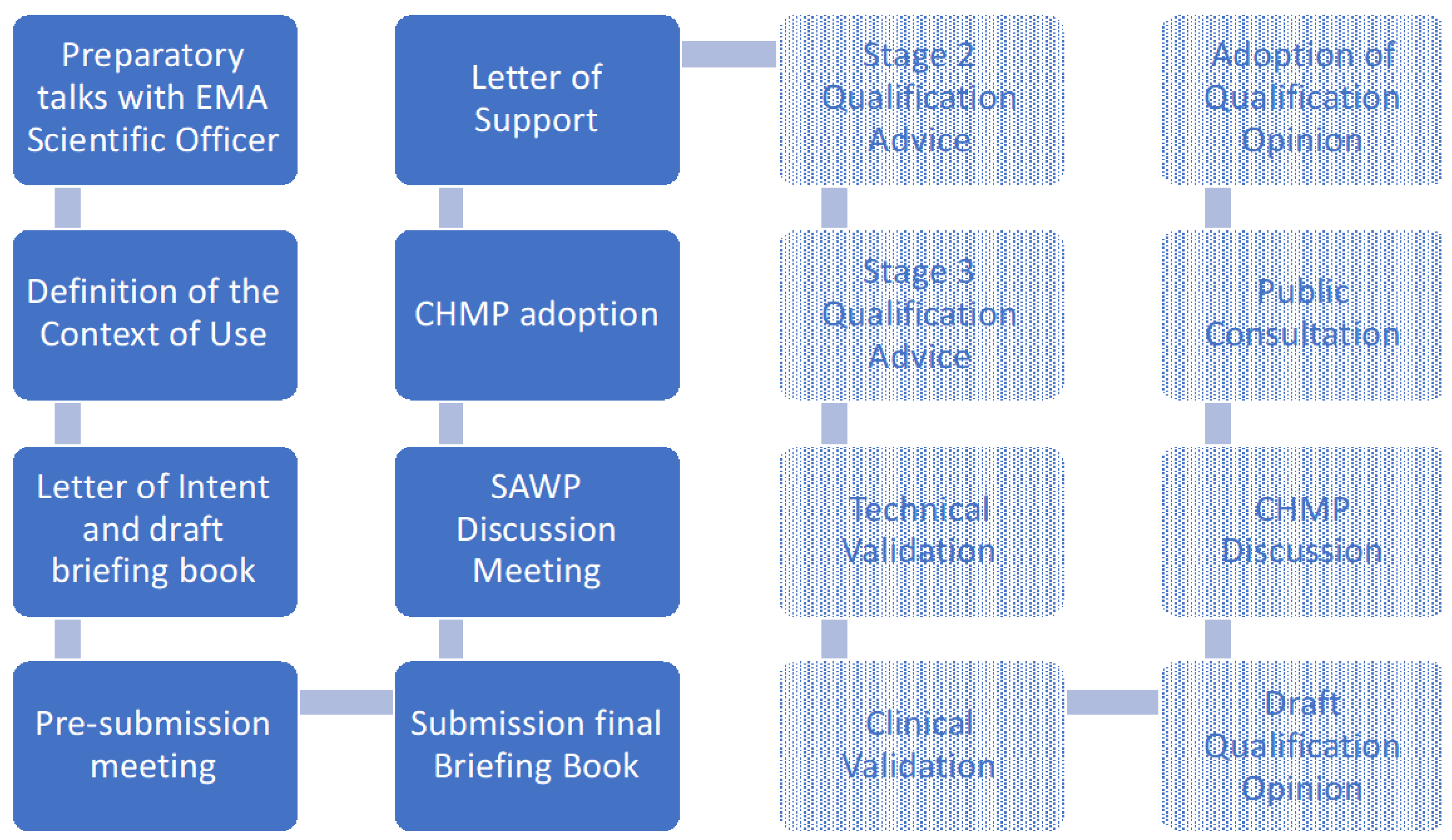
Sensors | Free Full-Text | Toward a Regulatory Qualification of Real-World Mobility Performance Biomarkers in Parkinson's Patients Using Digital Mobility Outcomes

GlaxoSmithKline's Juluca, Amgen's Repatha and more score EU backing in busy week for CHMP | Fierce Pharma

Unlocking the EMA Meetings: the Process and How to Prepare Adequately | Voisin Consulting Life Sciences

Daniele Focosi, MD PhD MSc on Twitter: "Another failure for molnupiravir…. @theosanderson @PeacockFlu @LongDesertTrain @OliasDave @siamosolocani @ACasadevall1 https://t.co/03A0Ipd8Cc" / Twitter

SERB reçoit un avis favorable du CHMP pour l'utilisation de Voraxaze® (glucarpidase) en tant que traitement d'urgence de la toxicité du méthotrexate liée à un retard d'élimination

Pfizer and BioNTech Receive Positive CHMP Opinion for Omicron BA.4/BA.5-Adapted Bivalent COVID-19 Vaccine Booster in European Union | Business Wire