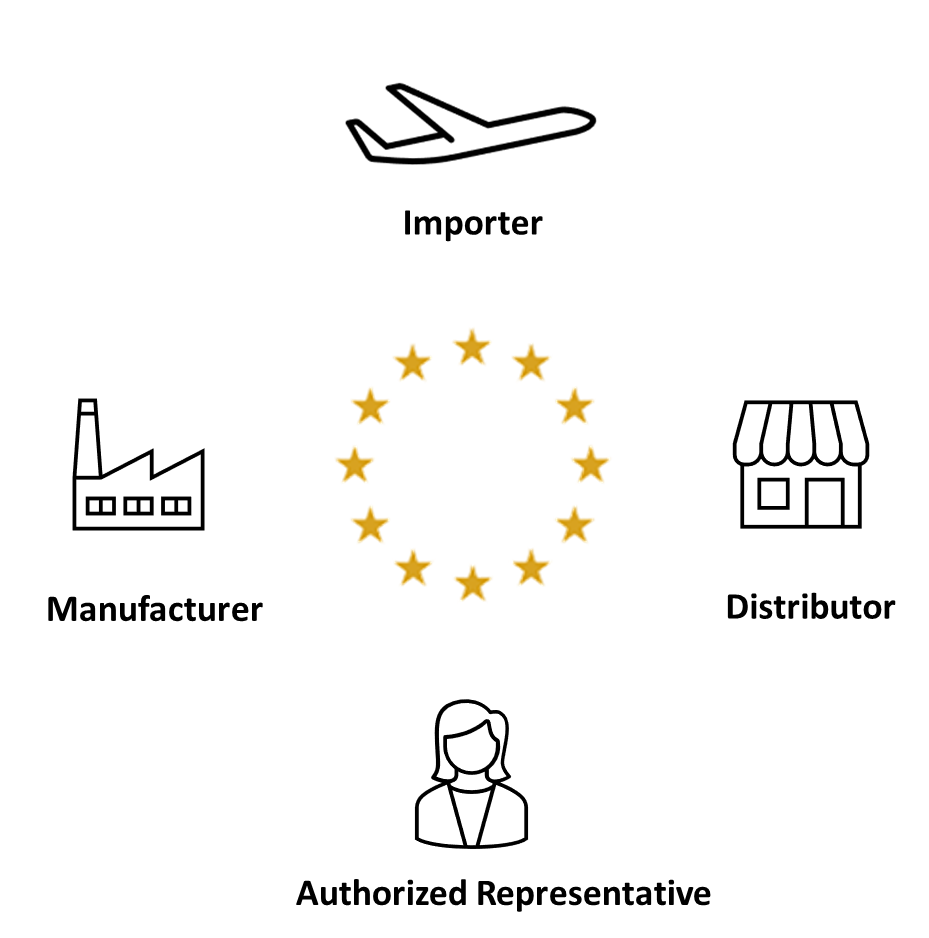
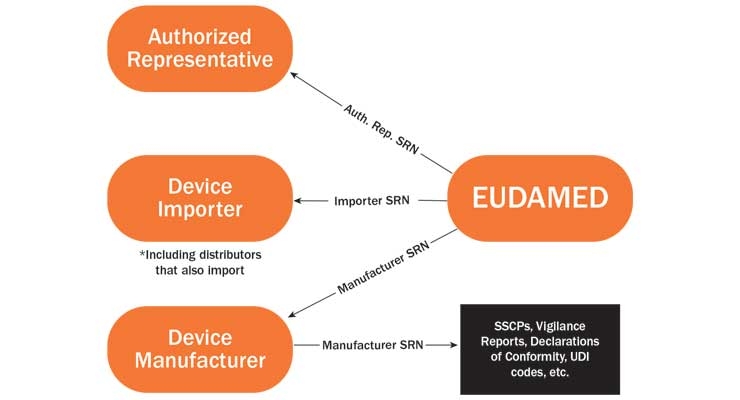
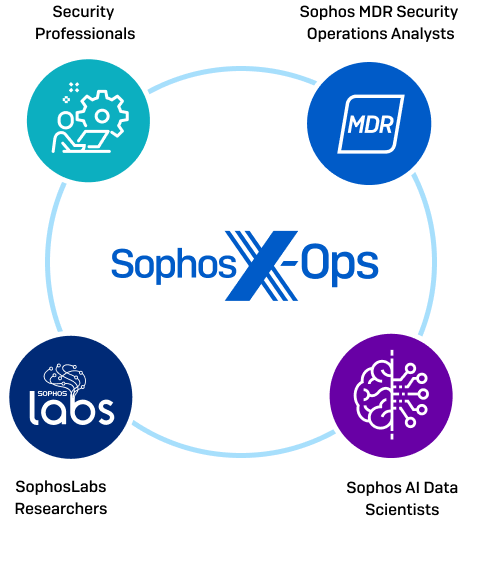
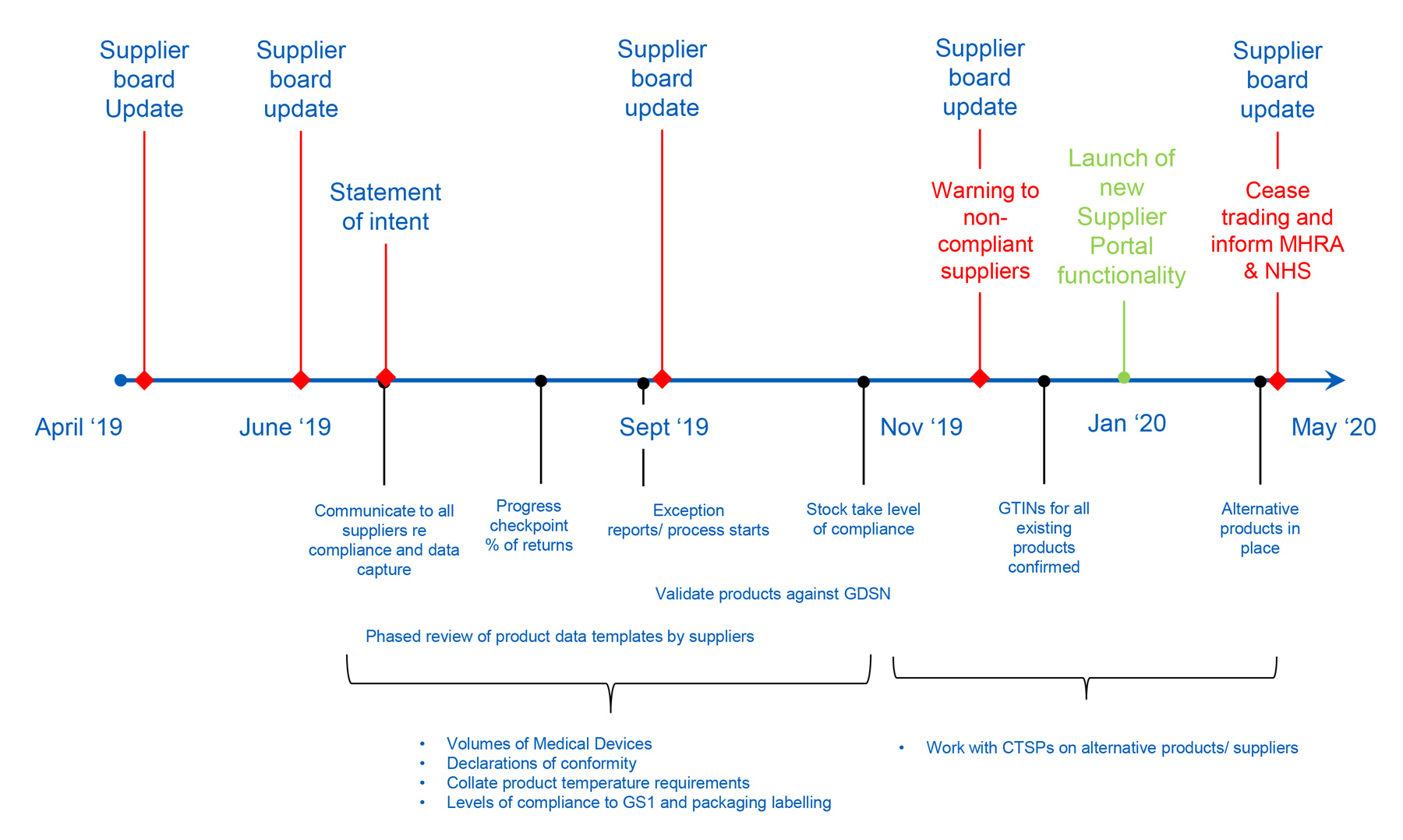
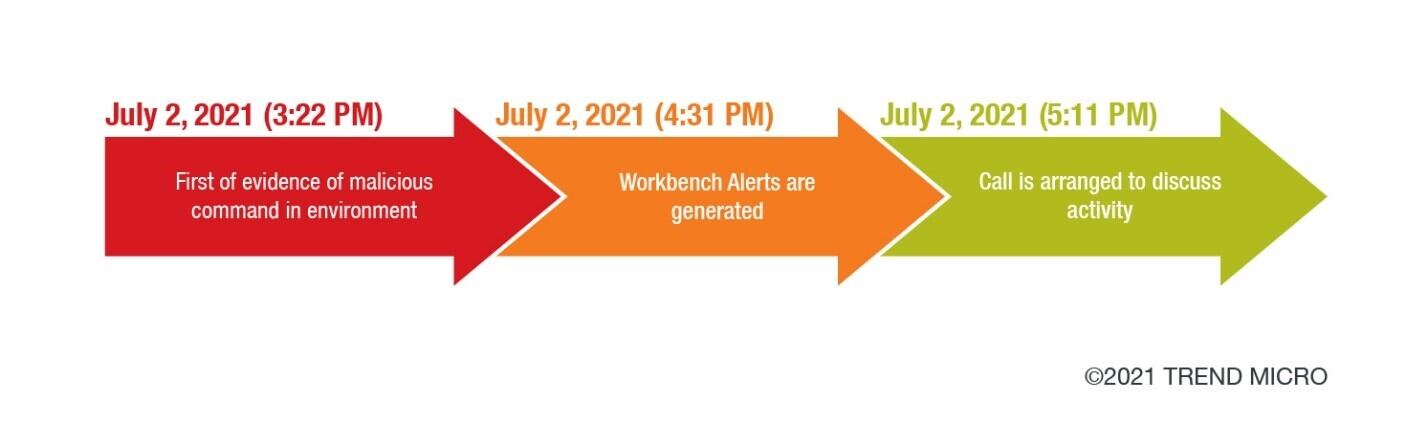
Emerson Automation Solutions - The Medical Device Regulation (MDR) will focus more on the supply chain of medical device manufacturers in the EU. This means that suppliers in the industry must have

Strengthening the public downstream SLD supply chain for MDR-TB : lessons learnt from a Western Cape case study | Semantic Scholar