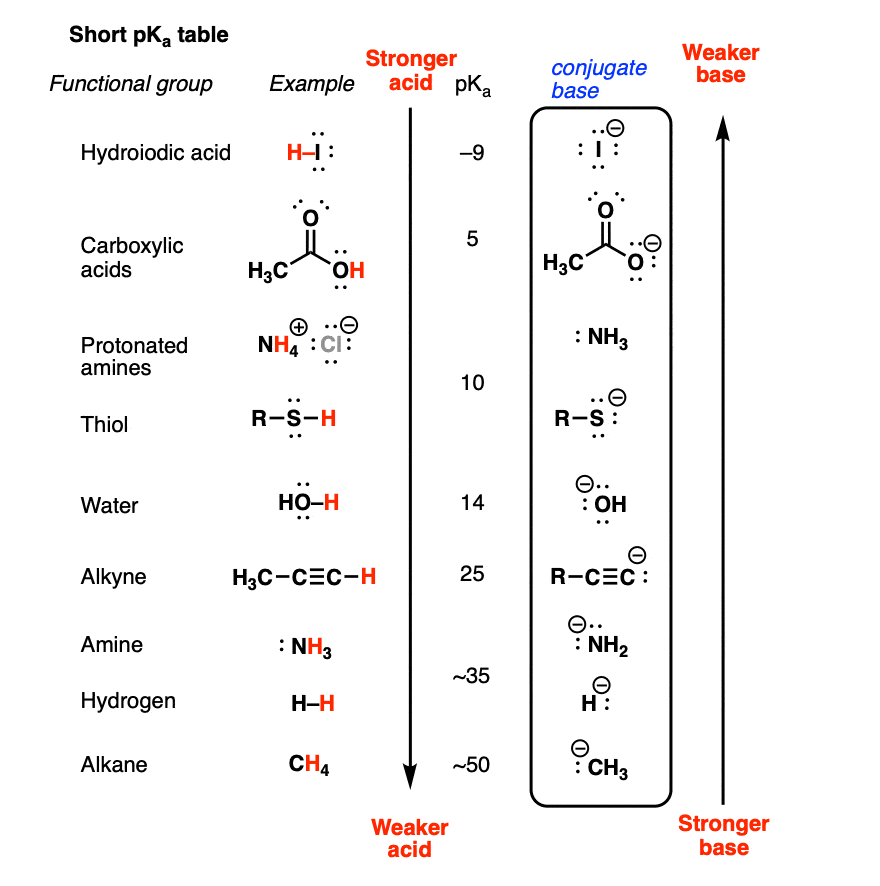
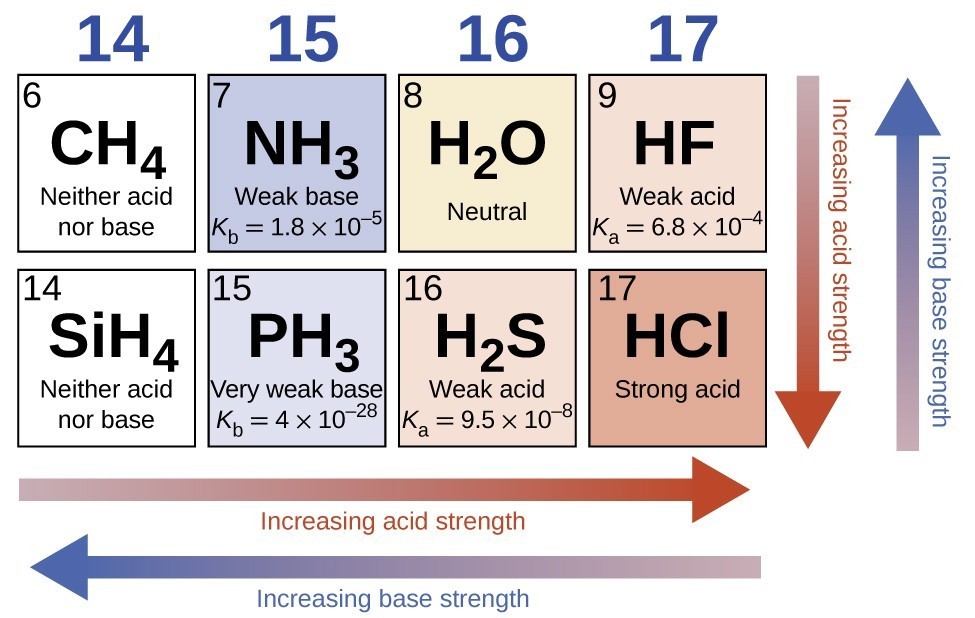
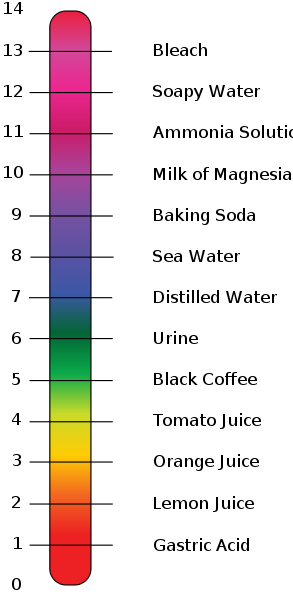
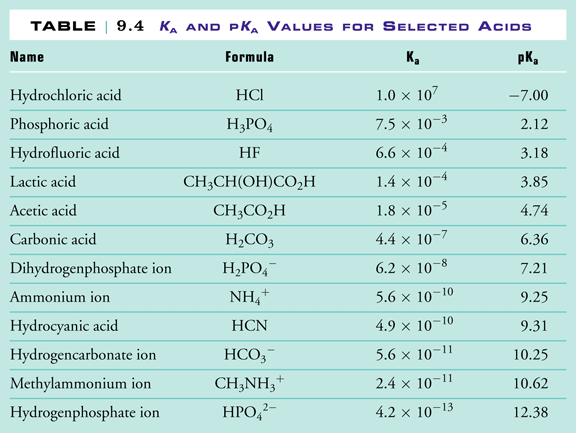
Use the Acid-Base Table to determine the of the weak acid Express your answer to two decimal places. | Homework.Study.com

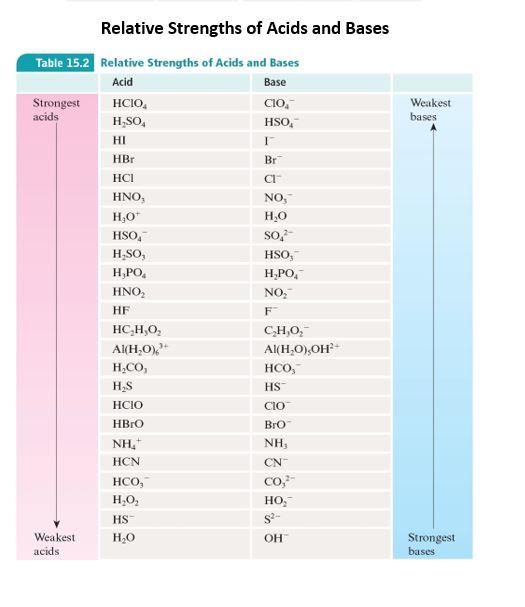
SOLVED: 4) Fill out the conjugate acids/bases in the following table: Conjugate Acid Base Pairs Acid: HF Conjugate base: Base: SOa" Conjugate acid: Base: Cl Conjugate acid: Acid: HCIOa Conjugate base: Acid:
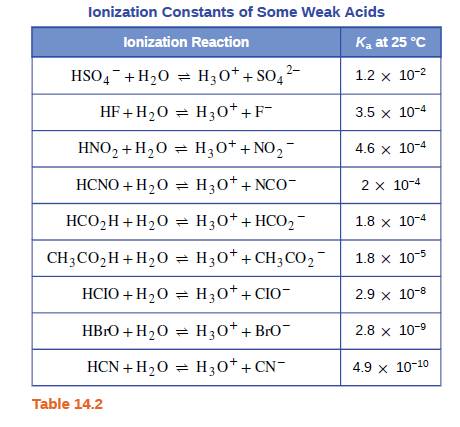
Which acid in Table 14.2 is most appropriate for preparation of a buffer solution with a pH of 3.7? Explain your choice. | bartleby