Development of Physiological Potency Assays for Cell-Based Therapies::Institute of Cell and Tissue Culture Technologies::Department of Biotechnology (DBT)::BOKU
The Challenges of Potency Assay Development for Cell-based Medicinal Products in Europe | Voisin Consulting Life Sciences

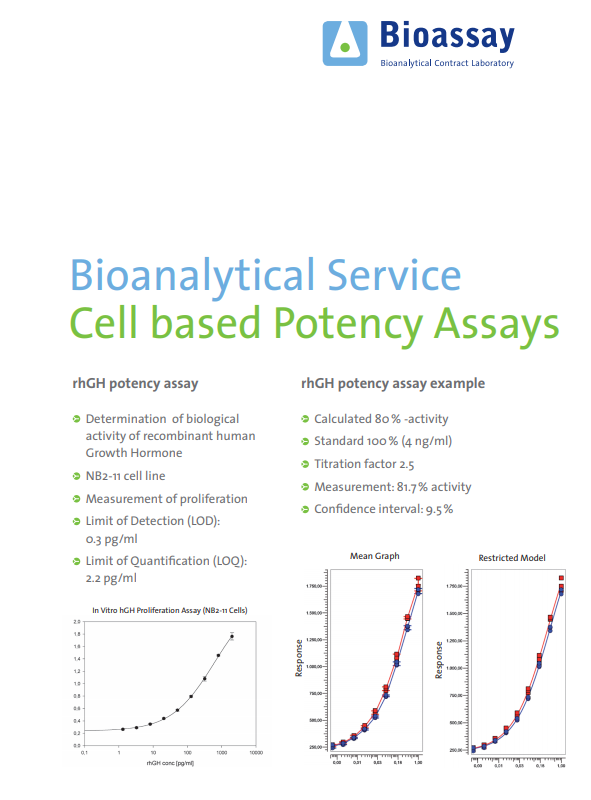
An ex vivo potency assay to assess active drug levels of a GLP-1 agonistic peptide during preclinical safety studies | Bioanalysis

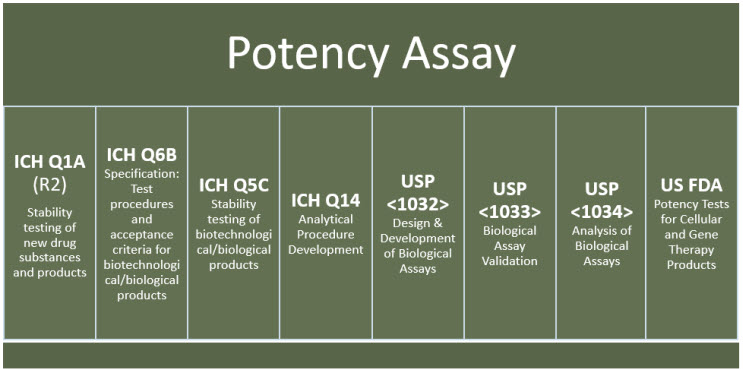
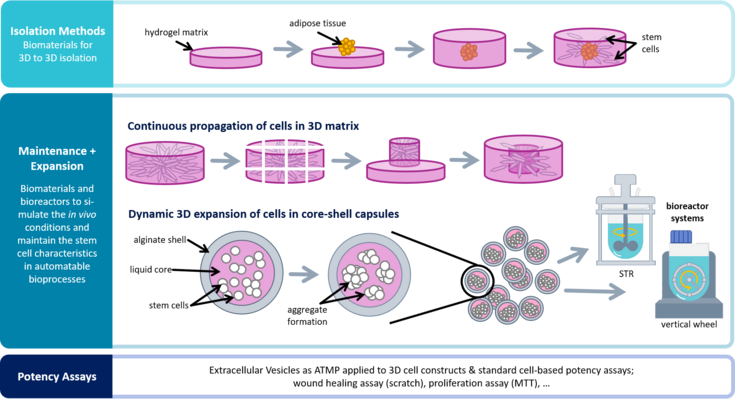
Potency assay development for cellular therapy products: an ISCT∗ review of the requirements and experiences in the industry - Cytotherapy

A Quantitative In Vitro Potency Assay for Adeno-Associated Virus Vectors Encoding for the UGT1A1 Transgene: Molecular Therapy - Methods & Clinical Development
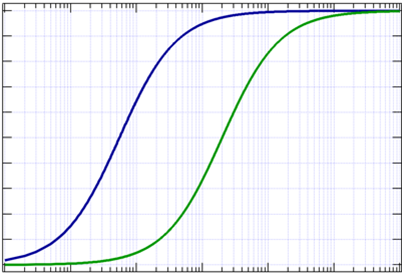
Cell-based potency assay (CBPA) comparison of incobotA (50 U, 100 U,... | Download Scientific Diagram

Mechanism of action of TDB1 and the reporter-gene potency assay. (a).... | Download Scientific Diagram
Botulinum Neurotoxin Serotype a Specific Cell-Based Potency Assay to Replace the Mouse Bioassay | PLOS ONE
Botulinum Neurotoxin Serotype a Specific Cell-Based Potency Assay to Replace the Mouse Bioassay | PLOS ONE