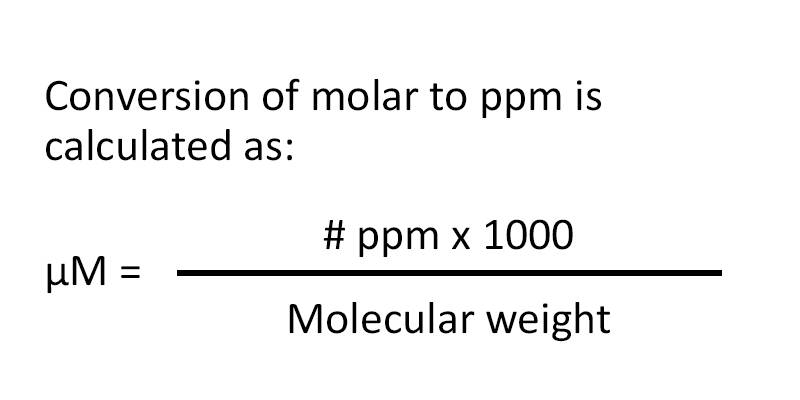
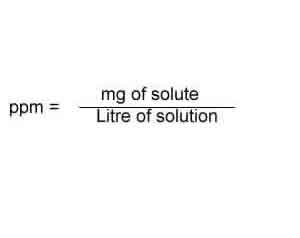
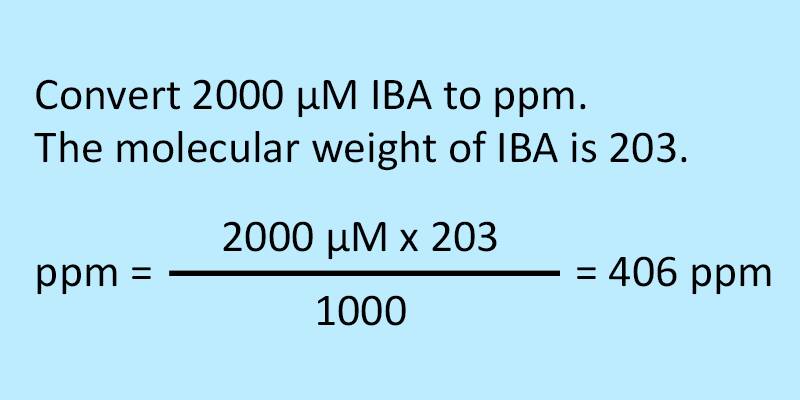
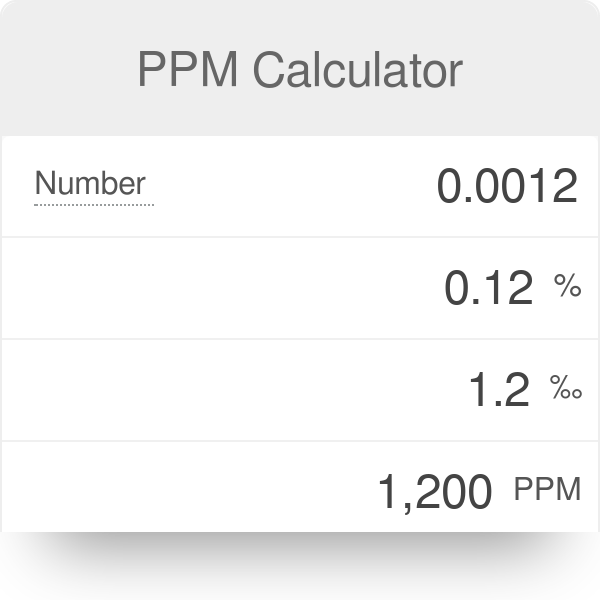
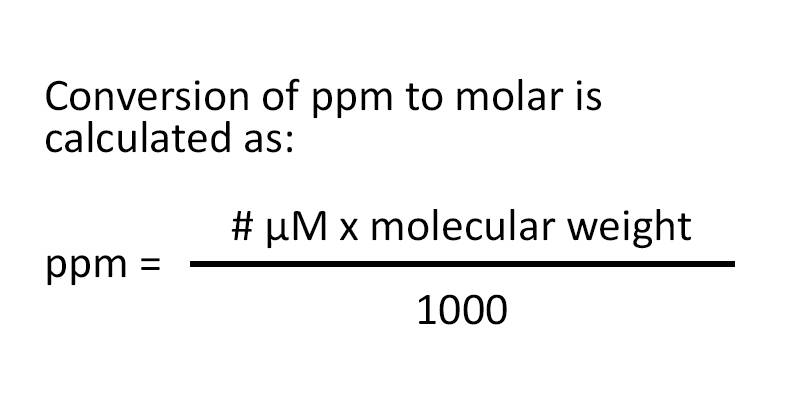
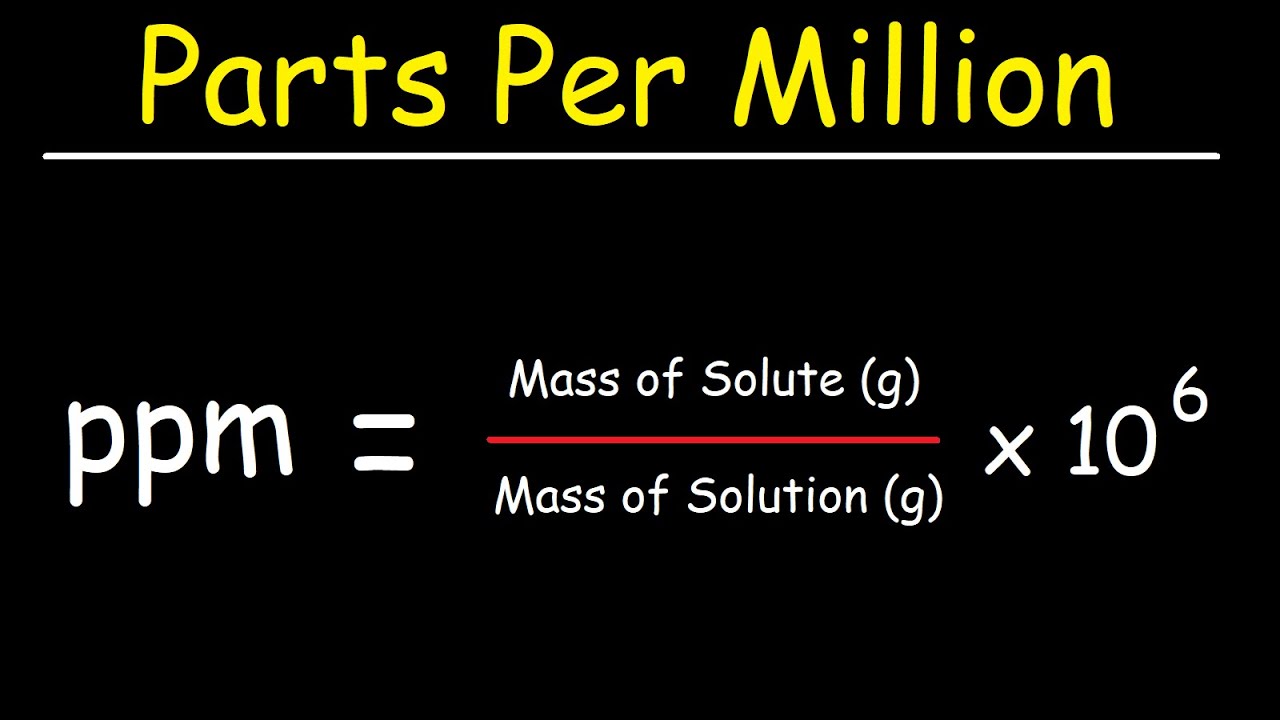SOLVED: Step 7 of 8 (b) Concentration of nitrite =1.74 ppm * 69.027 14.007 8.574 ppm This can be transformed into molar concentration as 8.574 ug 12 10 " 2 10' mL mol 8.574 ppm 1g JL 1ug TL 69.027 g I24xIO'M
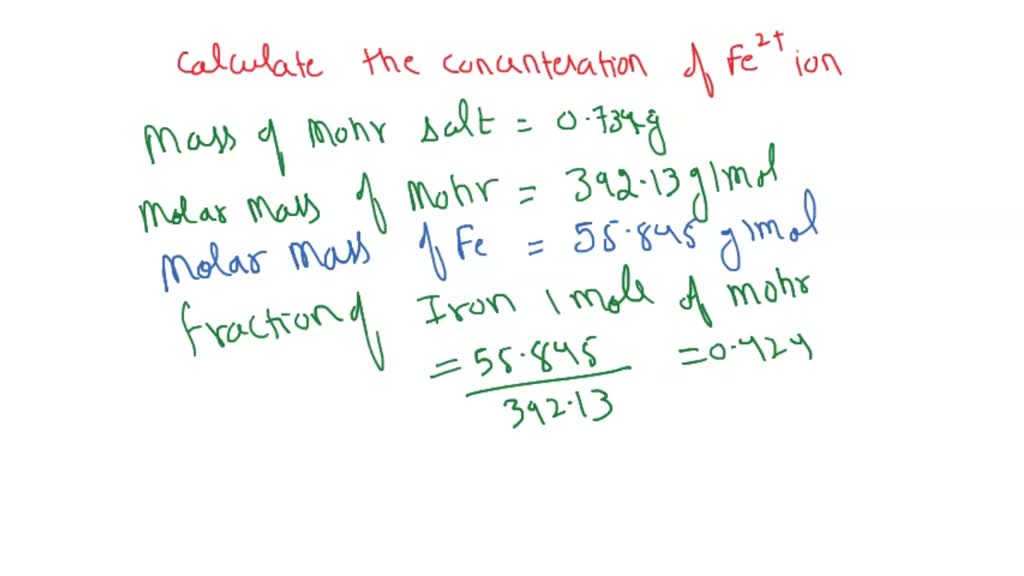
SOLVED: Calculate the theoretical concentration of Fe2+ in ppm (mg/L) if 0.734 grams of Fe(NHa)(SO4)2 6HzO salt was weighed on an analytical balance and dissolved in a 1000.0 mL volumetric flask which