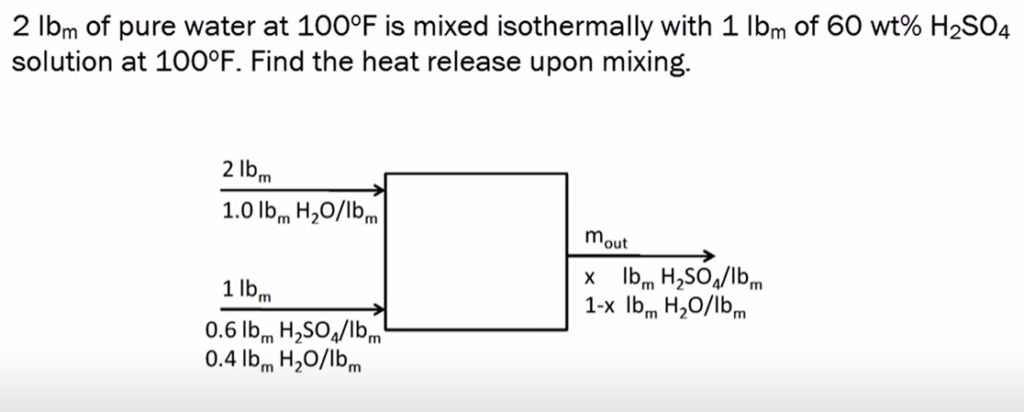
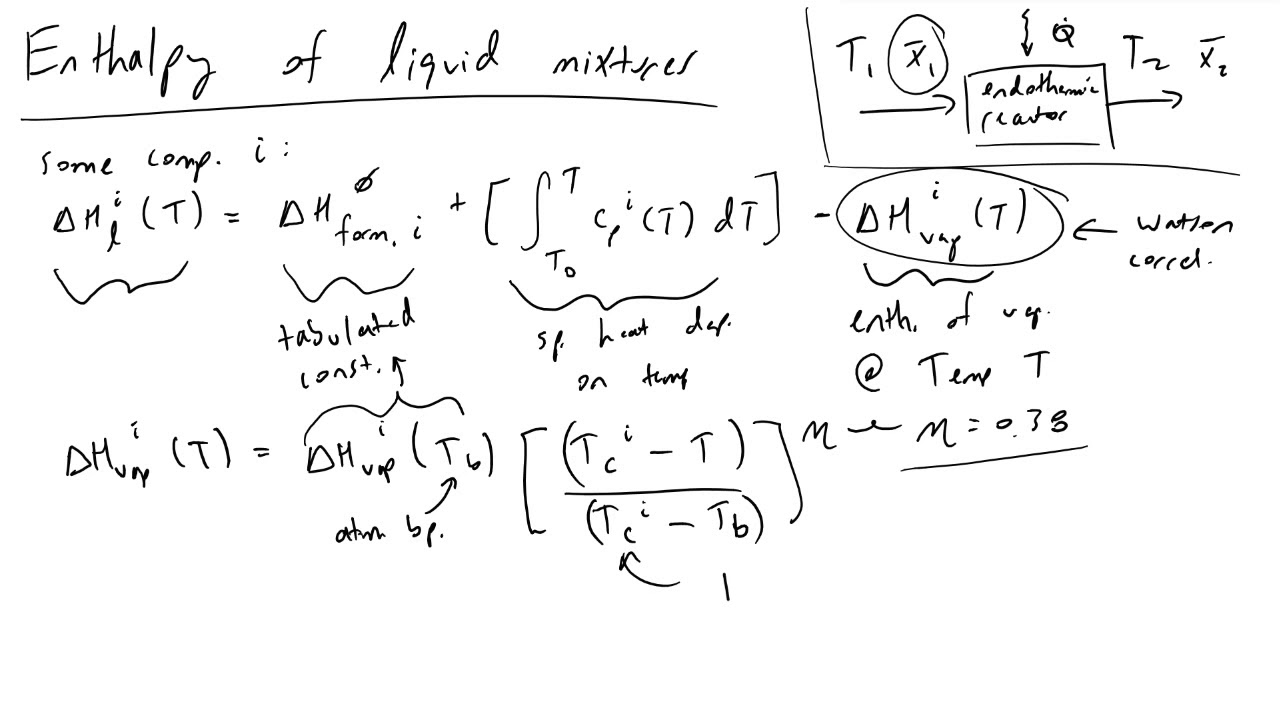
SOLVED: Consider a A-B binary solution. Given that the heat of mixing is AHmix XAXB(aXA + bXB), where a and b are constants, Compute the partial molar enthalpies AHA and AHB of

Free energy of mixing (a), enthalpy of mixing (b), entropy of mixing... | Download Scientific Diagram