Development of homogeneous plasmonic potency assay using gold nanoparticle immunocomplexes - ScienceDirect
Botulinum Neurotoxin Serotype a Specific Cell-Based Potency Assay to Replace the Mouse Bioassay | PLOS ONE

Critical considerations for the development of potency tests for therapeutic applications of mesenchymal stromal cell-derived small extracellular vesicles - Cytotherapy

An ex vivo potency assay to assess active drug levels of a GLP-1 agonistic peptide during preclinical safety studies | Bioanalysis

Development of an In Vitro Biopotency Assay for an AAV8 Hemophilia B Gene Therapy Vector Suitable for Clinical Product Release - ScienceDirect
![PDF] The challenges of potency assay development for cell-based medicinal products in Europe | Semantic Scholar PDF] The challenges of potency assay development for cell-based medicinal products in Europe | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/980465a72baf4590211336b890290a75eaffaf60/3-Figure2-1.png)
PDF] The challenges of potency assay development for cell-based medicinal products in Europe | Semantic Scholar

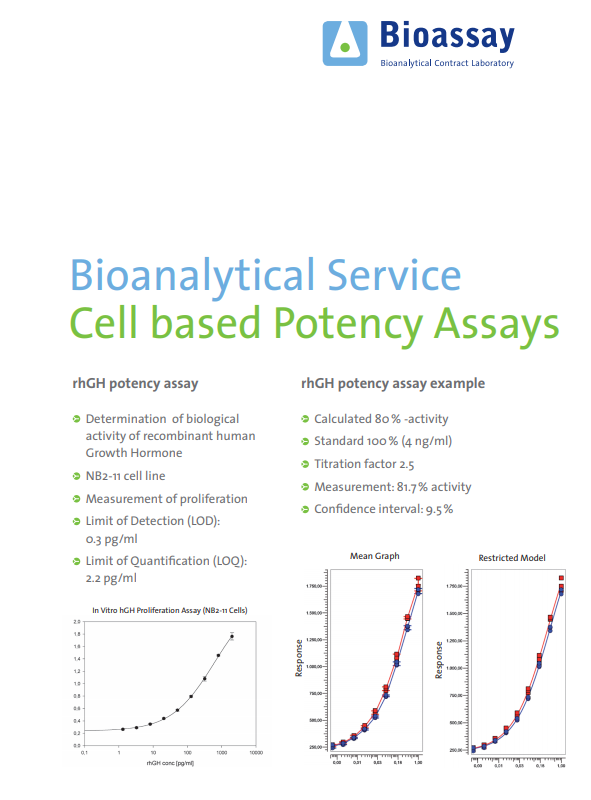
Accelerating Biologic and Biosimilar Drug Development: Ready-to-Use, Cell-Based Assays for Potency and Lot-Release Testing - BioProcess InternationalBioProcess International

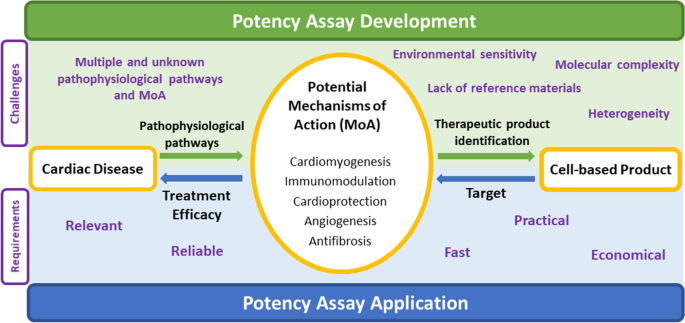
The Essential Need for a Validated Potency Assay for Cell-Based Therapies in Cardiac Regenerative and Reparative Medicine. A Practical Approach to Test Development | SpringerLink
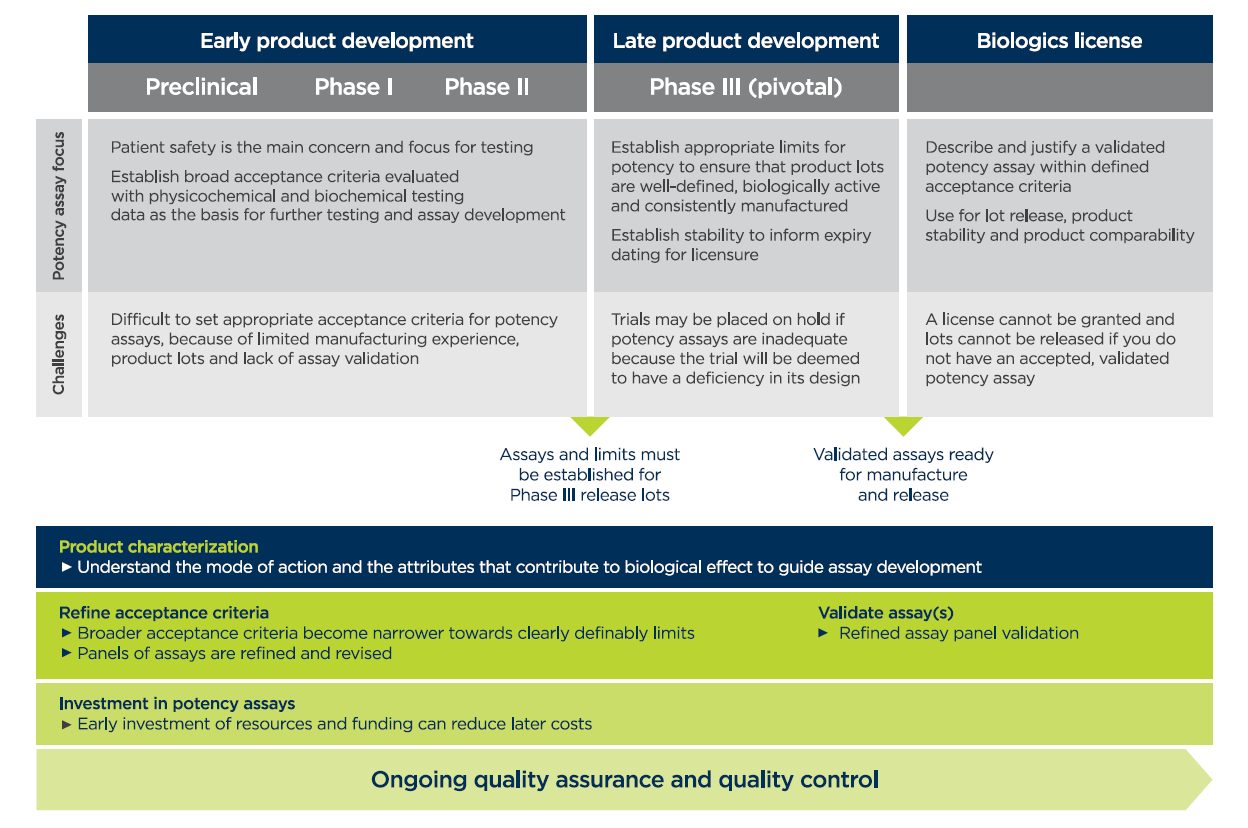
Potency assays for ATMPs: overcoming challenges on the path to commercialization - Insights From Our Labs to Yours

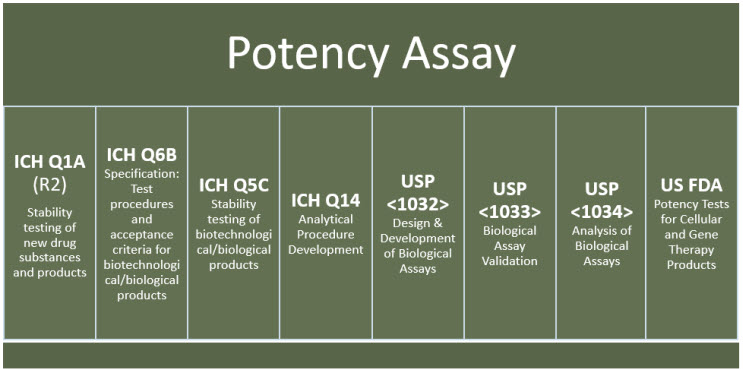
Potency assay development for cellular therapy products: an ISCT∗ review of the requirements and experiences in the industry - Cytotherapy